[ad_1]
ktsimage
Poseida Therapeutics brand (Poseida Thereapeutics web site)
San Diego-based Poseida Therapeutics, Inc. (NASDAQ:PSTX) is a clinical-stage biopharmaceutical firm that has developed a collection of proprietary genetic engineering platform applied sciences to create differentiated cell and gene therapeutics with the potential to treatment cancers, uncommon ailments and past with a single remedy with minimal uncomfortable side effects.
Poseida’s market cap of roughly $300 million is the same as its present estimated $300 million in money (or equivalents), which features a current $110 million obtained from Roche described in additional element beneath.
For the explanations mentioned on this article, the writer believes Poseida is at the moment undervalued.
Poseida’s Three Platforms of Genetic Engineering (Poseida’s August 2022 Presentation)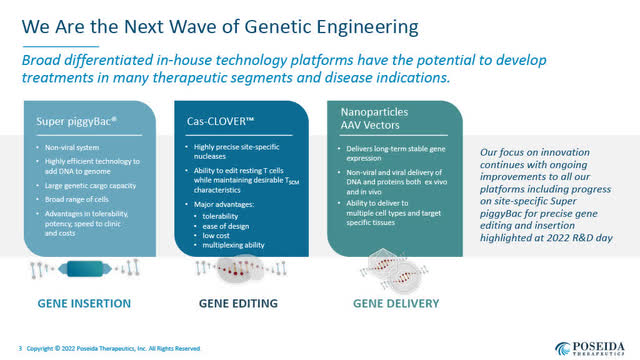
Poseida’s three central internally developed expertise platforms embody:
Tremendous piggyBac DNA supply system (Poseida June 2022 company presentation)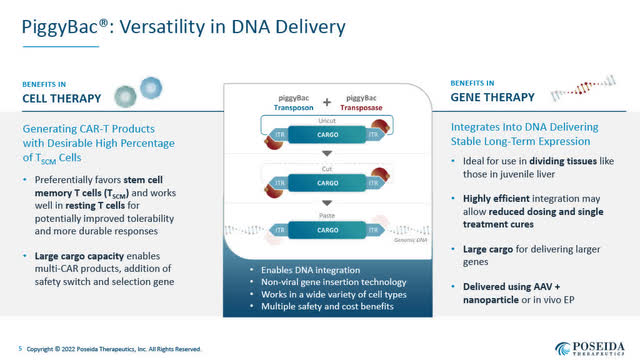
- a non-viral Tremendous piggyBac DNA supply system for gene insertion, which gives versatility in DNA supply;
- a non-viral Cas-CLOVER site-specific gene enhancing system; and
- a nanoparticle and AAV-based gene supply system.
Poseida believes its applied sciences present a differentiated and disruptive innovation in CAR-T cell and gene therapies with the potential to create single-treatment cures.
Whereas Poseida works with third events contract producers to provide its product candidates, it lately accomplished constructing its pilot GMP manufacturing facility in San Diego, California, adjoining to its headquarters to develop and manufacture preclinical supplies and medical provides for its allogeneic CAR-T Part 1 and a pair of medical trials. (Supply: Type 10-Q filed with the SEC on August 11, 2022).
Roche Strategic International Collaboration Settlement:
On August 3, 2022, Poseida introduced a strategic international collaboration settlement with F. Hoffmann La Roche Ltd. and Hoffmann-La Roche Inc. (collectively “Roche”) (OTCQX:RHHBY) (the “Roche Settlement”) probably value as much as $6 billion. The deal contains the fee of $110 million upfront, with a further $110 million in near-term milestone funds primarily based on what Poseida’s CEO describes as readily achievable targets that may seemingly be met.
The Roche Settlement turned efficient upon the expiration or termination of the relevant ready interval (usually 30 days) underneath the Hart-Scott-Rodino Antitrust Enhancements Act of 1975. Accordingly, it’s anticipated the Roche Settlement will shut and be efficient by the publication date of this text or shortly thereafter.
The Firm held an investor webcast on August 3 to debate the Roche deal. The webcast is on the market on the Firm’s web site right here.
$80,500,000 Observe-up Financing:
On August 4, Poseida introduced a $70,000,000 financing (20 million shares at $3.50) topic to a further 30-day 15% dealer/underwriter choices, all of which have been exercised, and which raised gross proceeds of $80,500,000 (roughly $75.3 Million internet after charges).
Poseida’s Prospectus Complement filed on August 4, 2022, with the SEC can be referred to beneath occasionally in addition to the Firm’s Type 10-Q filed with the SEC on August 11, 2022.
I additionally had a 30-minute Zoom name with Poseida’s CEO Mark Gergen on August 9 previous to writing this text.
Funding Abstract:
Poseida’s differentiated proprietary genetic engineering applied sciences are getting used to develop and advance next-generation cell and gene therapeutics.
Poseida’s newly introduced international collaboration cope with Roche (regarding its present and extra next-generation allogeneic CAR-T cell therapies within the remedy of hematologic malignancies, probably value $6 billion and as much as $220 million upfront or close to time period), follows the announcement in October 2021 of a significant Takeda (TAK) collaboration deal value as much as $3.6 billion, regarding Poseida’s gene remedy expertise.
The Roche and Takeda agreements assist validate the science, worth and potential of Poseida’s expertise platforms and considerably defray Poseida’s future medical trial and improvement prices.
Poseida’s market cap of roughly $300 million is the same as the estimated $300 Million worth of its money (or equivalents) which Poseida is anticipated to have in early September. The writer believes that the present market cap undervalues Poseida’s cell and gene platform applied sciences, a number of cell and gene remedy medical applications, pre-clinical pipeline, mental property and money.
There seems to be a disconnect in how the scientific and business neighborhood (as validated by the current Roche Settlement and the Takeda Settlement) views the worth of Poseida versus how the market is valuing the Firm.
Poseida’s upcoming catalysts embody two near-term H2 readouts in two Part 1 medical trials in addition to the probability of receiving a further $110 million in near-term milestone funds from Roche (which Poseida signifies are milestones which are extremely prone to be met inside a yr, if not sooner).
Whereas Poseida has nice promise, it stays an early-stage, high-risk firm, appropriate for risk-tolerant traders.
Poseida Therapeutics, Inc.: Company Abstract
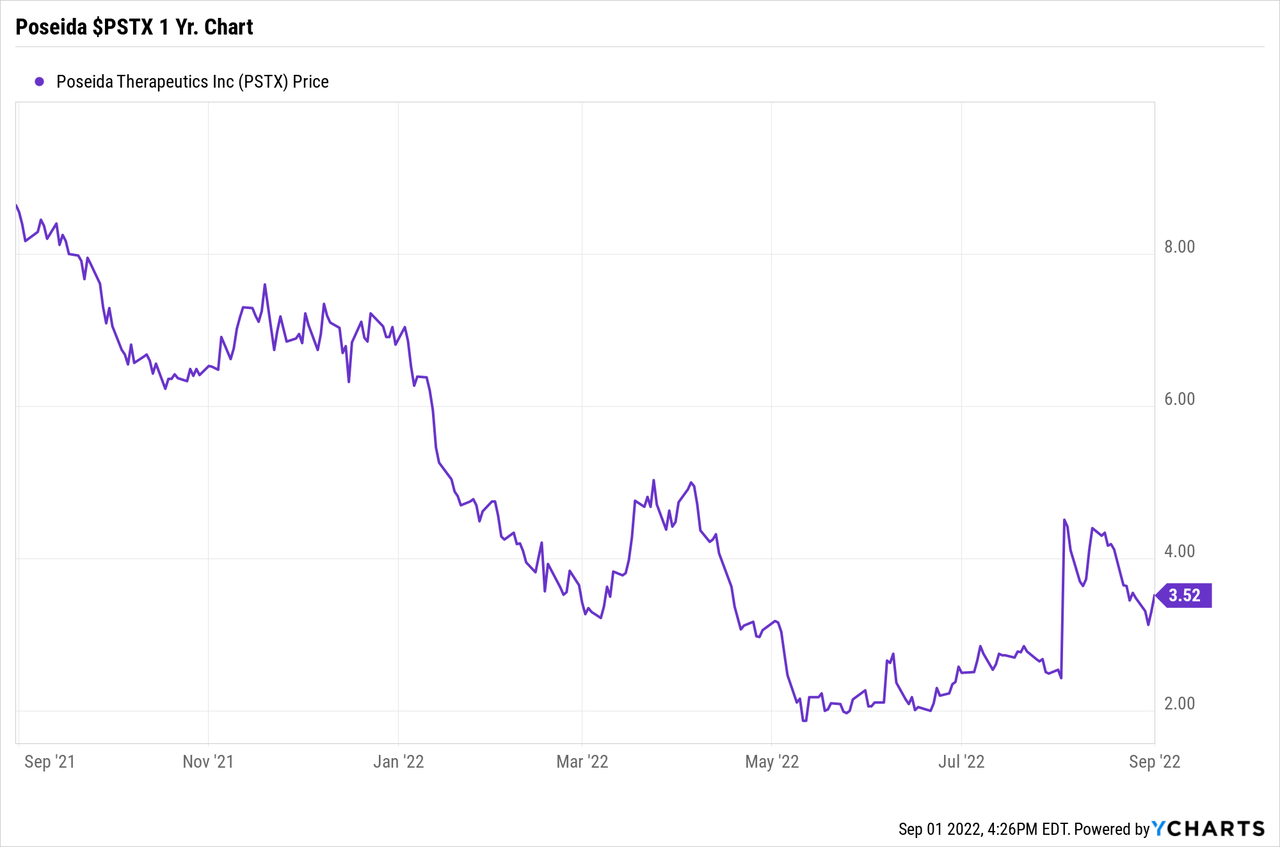
1 12 months Share Value Vary: $1.82 to $8.65
Share Value: Shut on September 1, 2022: $3.52
Market Cap, shut on September 1, 2022: Roughly $300 Million
85,728,726 widespread shares excellent following current financing: $80,500,000 gross proceeds, or approx. $75.3 million internet after charges. There are 85,728,726 widespread shares excellent following the current $80,500,000 financing. (Supply: Prospectus Complement)
The 85,728,726 shares don’t embody the 12,578,225 excellent choices (weighted common train worth of $8.06), 2,275,267 restricted inventory items, 121,122 warrants ($4.96 common train worth), 2,614,787 shares that may be issued underneath the 2020 Fairness Incentive Plan, and 1,621,649 shares reserved for future issuance underneath Worker Inventory Buy Plan. (Supply: Prospectus Complement)
Unaudited money and equivalents as of June 30, 2022: approx. $142.6 million (Supply: Prospectus Complement)
Money (estimated by the author) as of early September 2022: together with the $110 million upfront obtained from Roche and the online $75.3 million obtained from this month’s financing, I’ve estimated Poseida’s present money place at roughly $300 million (my estimate).
Money Burn: Based on its Type 10-Q, filed with the SEC on August 11, 2022, Poseida utilized internet money of $91.1 million throughout the 6 months ending June 30, 2022. Beneath the phrases of the Roche Settlement, Roche will assume sure medical trial prices sooner or later however I’m assuming that the money burn price will proceed at its current price for the subsequent few years. The Firm has offered steering that conservatively, it at the moment has adequate liquidity to hold out its present plans into mid-2024 with out making an allowance for a lot of the second anticipated $110 million in milestone funds that may be earned within the close to time period underneath the Roche Settlement, or together with some other milestone funds underneath both the Roche or Takeda agreements.
Time period Debt: Based on the Firm Type-10Q, Poseida entered right into a Mortgage and Safety Settlement with Oxford Finance LLC, as amended in February 2022 underneath which the Firm borrowed $60 million in time period loans. Curiosity-only funds are payable by way of to April 1, 2025, adopted by 23 equal month-to-month funds of principal and relevant curiosity, and different phrases. The mortgage could also be repaid on phrases previous to February 22, 2024, after which it might be repaid with none prepayment penalty.
Poseida’s Cell Remedy Pipeline:
Along with its gene remedy pipeline (mentioned later), Poseida’s Cell Remedy pipeline contains an autologous “auto” in addition to allogeneic “allo” chimeric antigen receptor T cell therapies, generally often called “CAR-T” therapies, within the remedy of varied cancers, together with strong tumors, a number of myeloma, B-Cell and Heme Malignancies.
For these unfamiliar with the phrases, “autologous” CAR-T are derived from the cells of the affected person being handled (being a customized and customarily very costly course of and probably deriving cells from a sick and infrequently aged particular person), whereas an “allogeneic” CAR-T is derived from a third-party donor’s cells which could be replicated making it a less expensive “off the shelf” product, and customarily derived from a youthful wholesome donor.
Poseida is concentrated on advancing its allogeneic CAR-T therapies the place it believes the way forward for CAR-Ts lie, though it started medical improvement with an autologous CAR-T cell program, P-PSMA-101, mentioned in additional element beneath, which Poseida believes has offered substantial learnings.
Poseida’s Differentiated Know-how:
As Poseida’s CEO Mark Gergen mentioned throughout the August 3 webcast, one in all Poseida’s differentiators is its deal with Tscm cells, in any other case often called stem cell reminiscence T cells, which Poseida believes are “the perfect cell sort for allogeneic cell therapies.”
Poseida believes it has proven that its proprietary non-viral Cas-CLOVER gene enhancing expertise is a superior technique of manufacturing CAR-T cell therapies which use T cells which are genetically modified.
In a peer-reviewed paper revealed June 29, 2022 within the Journal of Molecular Remedy Nucleic Acids (authored by members of the Poseida group, together with founder Dr. Eric Ostertag, MD, Ph.D.), Cas-CLOVER is a novel high-fidelity nuclease for secure and strong technology of TSCM-enriched allogeneic CAR-T cells, Poseida’s Cas-CLOVER gene enhancing has been proven to protect a better share of Tscm cells, which have a self-renewing capability, low off-target exercise, and a more practical anti-tumour response.
As set out within the paper’s summary,
The usage of T cells from wholesome donors for allogeneic chimeric antigen receptor T (CAR-T) cell most cancers remedy is enticing as a result of wholesome donor T cells can produce versatile off-the-shelf CAR-T remedies. To maximise security and sturdiness of allogeneic merchandise, the endogenous T cell receptor and main histocompatibility complicated class I molecules are sometimes eliminated through knockout of T cell receptor beta fixed (TRBC) (or T cell receptor alpha fixed [TRAC]) and B2M, respectively. Nevertheless, gene enhancing instruments (e.g., CRISPR-Cas9) can show poor constancy, which can lead to harmful off-target mutations. Moreover, many gene enhancing applied sciences require T cell activation, leading to a low share of fascinating stem cell reminiscence T cells (TSCM). We characterize an RNA-guided endonuclease, known as Cas-CLOVER, consisting of the Clo051 nuclease area fused with catalytically useless Cas9. In major T cells from a number of donors, we discover that Cas-CLOVER is a high-fidelity site-specific nuclease, with low off-target exercise. Notably, Cas-CLOVER yields environment friendly multiplexed gene enhancing in resting T cells. Along with the piggyBac transposon for supply of a CAR transgene in opposition to the B cell maturation antigen (BCMA), we produce allogeneic CAR-T cells composed of excessive percentages of TSCM cells and possessing potent in vivo anti-tumor cytotoxicity.”
Poseida’s Three Ongoing Part 1 CAR-T Scientific Trials
As proven within the Cell Remedy Pipeline slide beneath (additionally described in Poseida’s Prospectus Complement), Poseida at the moment has three ongoing Part 1 CAR-T medical trials:
P-PSMA-101 Interim Ph 1 Scientific Outcomes (Poseida June 2022 Presentation)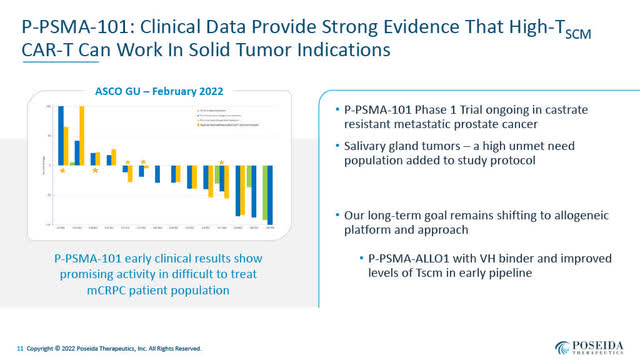
- P-PSMA-101, an autologous CAR-T concentrating on prostate-specific membrane antigen “PSMA” developed to deal with metastatic castrate-resistant prostate most cancers “mCRPC” and salivary gland carcinoma. Encouraging preliminary Part 1 ends in mCRPC have been introduced by Dr. Susan Slovin of Memorial Sloan Kettering Most cancers Heart on February 17, 2022 at ASCO-GU. A brief video of Dr. Slovin’s ASCO-GU presentation of these outcomes and describing P-PSMA-101 is on the market right here. A slide presentation above summarized the interim Part 1 medical knowledge introduced at ASCO-GU. Poseida signifies that additional medical outcomes from this P-PSMA-101 Part 1 trial will seemingly be introduced in 2023. (Observe: A second-generation allogeneic CAR-T program, P-PSMA-ALL01, concentrating on PSMA is at the moment in pre-clinical improvement).
- P-BCMA-ALL01, a absolutely allogeneic CAR-T concentrating on BMCA developed to focus on relapsed/refractory a number of myeloma. Preliminary medical outcomes are anticipated to be launched in H2 2022, topic to coordination with Roche. P-BCMA-ALL01 can be solely licensed to Roche underneath the newly introduced Roche settlement. Beneath the Roche settlement, Roche can be accountable for almost all of improvement prices for P-BCMA-ALL01 and can assume all improvement prices following the completion of the Part 1 trial.
- P-MUC1C-ALL01, a absolutely allogeneic CAR-T to deal with strong most cancers tumours, stays absolutely owned by Poseida. Preliminary Part 1 outcomes are anticipated to be launched in H2 2022 and if profitable, will assist validate Poseida’s thesis of creating CAR-Ts with a bigger proportion of Tscm T-cells to soundly and successfully deal with strong tumours.
Poseida’s International Collaboration Settlement with Roche Centered on Allogeneic CAR-T Cell Therapies for Hematologic Cancers Introduced August 3, 2022
Poseida introduced a strategic international collaboration and licensing settlement with Roche (OTCQX:RHHBY) to advance various Poseida’s present and extra next-generation allogeneic CAR-T cell therapies within the remedy of hematologic malignancies.
Poseida retained full rights to its allogeneic CAR-T cell applications in strong tumours.
Poseida’s International Partnership with Roche (Poseida August 2022 company presentation)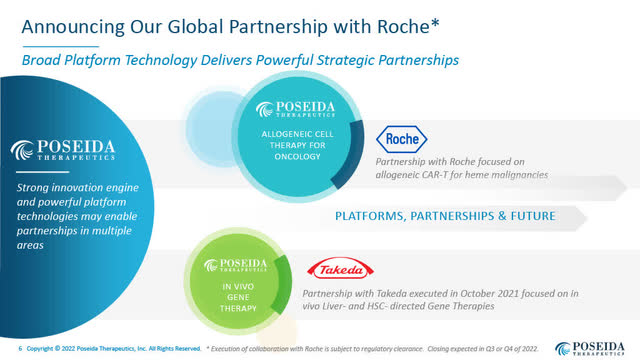
Beneath the Roche Settlement, Poseida will obtain $110 million upfront and as much as a further $110 million in near-term milestones, which Poseida believes is very achievable. In my current dialog with Mark Gergen, he confirmed that by “near-term” he meant that the $110 million in milestones would extremely seemingly be achieved inside roughly 12 months.
As well as, Poseida is eligible for extra internet milestones and different funds from Roche probably value as much as $6 billion in worth, in addition to tiered royalties into the low double-digits on internet gross sales royalties.
In return, Roche will get entry to various “off-the-shelf” allogeneic CAR-Ts for hematologic malignancies, together with P-BCMA-ALLO1 for a number of myeloma, (at the moment in a Part I examine), and P-CD19CD20-ALLO1, described as an allogeneic “twin CAR-T” for B-cell malignancies, for which an IND is anticipated to be filed in 2023.
Poseida and Roche can even work on extra targets.
Financial Abstract of Roche Settlement (Poseida August 3, 2022 presentation)
Strategic Rationale for Roche Settlement (Poseida August 3, 2022 presentation)
Collaboration Scope of Roche Settlement (Poseida August 3, 2022 presentation)
October 2021: Poseida – Takeda Collaboration Settlement in Gene Remedy
The Roche deal follows a significant analysis collaboration and licensing deal in In-Vivo gene remedy that Poseida entered into with Takeda Pharmaceutical Firm Ltd. (TAK), introduced on October 12, 2021.
The Takeda settlement offered Poseida with $45 million upfront and probably as much as $3.6 billion in funds if all choices are exercised and milestones achieved, along with tiered royalties on industrial gross sales.
Strategic Partnership with Takeda (Poseida August 3, 2022 presentation)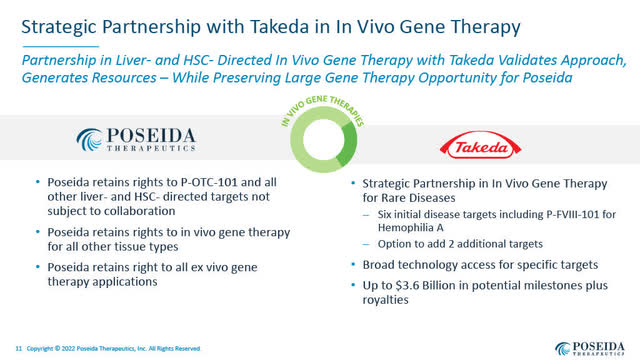
The Takeda settlement permits Takeda,
…to make the most of Poseida’s piggyBac, Cas-CLOVER, biodegradable DNA and RNA nanoparticle supply expertise and different proprietary genetic engineering platforms for the analysis and improvement of as much as eight gene therapies”.
Poseida’s Senior Management Workforce:
Poseida has an skilled administration group led by Mark Gergen, appointed CEO in February 2022. Previous to that date, Mark Gergen was Poseida’s President and Chief Enterprise Officer. He has been a director since 2018. Previous to Poseida, he was Senior Vice President and Chief Working Officer of Halozyme, Inc. and Government Vice President and Chief Working Officer at Mirati Therapeutics (MRTX). (For extra details about Poseida’s management group, see Poseida’s web site)
Administrators:
Poseida has a bunch of seven very skilled Administrators. Their names and biographies could be discovered on Poseida’s web site right here. Of explicit observe, Along with Mark Gergen, Poseida’s Administrators embody:
Government Chairman Dr. Eric Ostertag, M.D., Ph.D., who served as Poseida’s founding CEO from July 2015 to January 2022. Based on Poseida’s web site, Dr. Ostertag invented or co-invented a majority of the genetic engineering platform applied sciences utilized by Poseida for human therapeutics.
Dr. Charles Baum, President, Head of Analysis and Growth, and a Director at Mirati Therapeutics (MRTX), an organization he based and the place he served as CEO from 2012 to 2021.
Ms. Cynthia Collins, with greater than 40 years of expertise in cell and gene therapies and the life sciences, served because the CEO of Editas Drugs (EDIT), a number one pioneer in gene enhancing.
Poseida’s Scientific Advisory Board additionally contains a powerful group.
Upcoming Catalysts
Poseida’s upcoming milestones and catalysts embody:
- Part 1 Scientific replace from P-BCMA-ALL01, absolutely allogeneic CAR-T concentrating on BMCA developed to focus on relapsed/refractory a number of myeloma in H2 2022 (though the disclosure can be topic to coordination with Roche);
- Part 1 Scientific replace from P-MUC1C-ALL01, absolutely allogeneic CAR-T to deal with strong most cancers tumours, which stays absolutely owned by Poseida in H2 2022;
- Part 1 medical outcomes from P-PSMA-101, autologous CAR-T, within the remedy of mCRPC in 2023;
- Second-generation pre-clinical allogeneic CAR-T program, P-PSMA-ALL01, concentrating on PSMA; IND submitting and initiation of Part 1 medical trial in 2023 (disclosure topic to coordination with Roche);
- Further milestone funds of $110 million from Roche in H2 2022 and 2023 (my estimate primarily based on administration’s recommendation that the milestones are simply achievable inside roughly the subsequent 12 months).
Analysts’ Value Targets
Agency: Suggestion Value Goal Date
BTIG (Justin Zelin) Purchase $20.00 Aug. 18, 2022
Cantor Fitzgerald (Jennifer Kim) Obese $24.00 Aug. 12, 2022
William Blair (Raju Prasad) Outperform No PT Aug. 11, 2022
Piper Sandler (Edward Tenthoff) Obese $11.00 Aug. 11, 2022
BofA Securities (Alec Stranahan) Purchase $12.00 Aug. 3, 2022
Abstract
Each the lately introduced Roche Settlement and final October’s Takeda Settlement have helped validate the science and potential of Poseida’s differentiated proprietary genetic engineering applied sciences in addition to a few of its medical stage and early-stage pipelines.
Poseida’s proprietary applied sciences embody:
1. non-viral Tremendous piggyBac DNA supply system for gene insertion;
2. non-viral Cas-CLOVER site-specific gene enhancing system; and its
3. nanoparticle and AAV-based gene supply system
Poseida’s near-term catalysts embody its two near-term H2 medical Part 1 readouts, and third Part 1 medical outcomes from its autologous drug candidate in 2023.
Along with its present money place, there’s additionally the probability of receiving a further $110 million in near-term milestone funds from Roche (which Poseida signifies are extremely prone to be met and the quantities paid inside roughly 12 months).
With a market cap of roughly $300 million, equal to the estimated $300 Million worth of its money (or equivalents), Poseida’s cell and gene platform applied sciences, a number of cell and gene remedy medical applications, pre-clinical pipeline, mental property and money are undervalued.
There seems to be a disconnect in how the scientific and business neighborhood (as validated by the current Roche Settlement and Takeda Settlement) views the worth of Poseida’s applied sciences and pipeline belongings versus how the market is valuing the Firm.
Analysts protecting Poseida have a bullish view of Poseida’s potential.
For traders who’ve accomplished their due diligence and perceive that Poseida is an early-stage biotech firm with a excessive danger/ excessive reward profile, Poseida could also be a pretty funding, notably for these with a 2 to 4-year funding time horizon.
Dangers
Poseida needs to be thought-about a high-risk, early-stage firm that’s unlikely to generate materials revenues, not to mention earnings, for years to come back. Traders may lose some or most of their funding.
Whereas Poseida has indicated that it has adequate liquidity to fund its operations together with medical trials by way of to mid-2024, and can seemingly earn extra milestone funds underneath the Roche and/or Takeda agreements, there isn’t a assurance that it’ll have adequate funds longer-term to fund its operations.
For additional particulars of the potential dangers concerned, see the danger components set out within the firm’s most up-to-date public SEC filings accessible on the Firm’s web site and on EDGAR.
[ad_2]
Source link



